
Images Sarah Vaughan, text Genevieve Carbonatto
A 38 year old man presents to the Emergency department with shortness of breath which he has had for 4 weeks. His symptoms have worsened over the past 3 days. He can no longer go up 2 flights of steps to his apartment and he has noticed his ankles have been swelling. He is married and has had recent work stressors which he is unwilling to talk about. He denies smoking, or alcohol but after much discussion he admits to smoking 1 -2 pipes of methamphetamine a day. On examination his heart rate is 130/min, his BP 140/80, saturations 94% on RA, he has mild pitting ankle oedema and bibasal crepitations on chest examination.
His ECG

Sinus tachycardia , T wave inversion lateral leads
CXRay
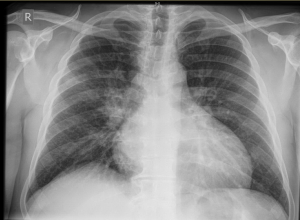
Cardiomegaly, globular heart
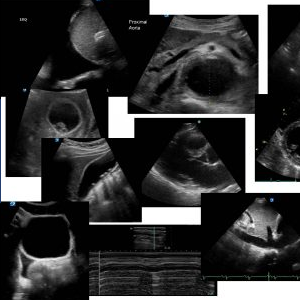
You decide to do an ECHO
This is his PLAX view
PLAX shows poor LV function, dilated RV and LA
This is his Apical 4 C view
The LV and RV are very poorly contracting and there is a thrombus visible at the apex of the LV. The RV is dilated.


This man has biventricular failure with biventricular dilatation the cause of which is not clear in the Emergency Department. The most common differential diagnosis for his cardiomyopathy is ischaemia, viral or drug induced cardiomyopathy. He had a normal angiogram and normal cardiac enzymes. The cause of his cardiomyopathy is metamphetamine use.
Discussion
There are 2 aspects of this case to discuss. Firstly cardiac complications of methamphetamines, secondly LV thrombus formation
The following is an excellent article published in April 2016 on the Cardiac Complications of Methamphetamines, by Elizabeth Raratz and Neil Cunningham(http://www.heartlungcirc.org/article/S1443-9506(15)01489-4/pdf)
The following is a brief summary of a small section of this superb article:
- Cardiovascular complications are the second leading cause of death in methamphetamine users (14%)
- 54% of methamphetamine users exhibit cardiovascular complications (mean age of users 32 years, 78% male)
- The cardiac complications of methamphetamines are hypothesised to arise from a variety of mechanisms. In animal studies, administration of methamphetamine to rats causes hypertension, tachycardia and myocardial toxicity with cellular death, fibrosis and contraction band necrosis (4). In human autopsy specimens, severe interstitial fibrosis and scar formation has been documented (5)
Methamphetamine cardiovascular pathologies include
- malignant hypertension
- aortic dissection
- acute coronary syndromes
- pulmonary arterial hypertension
- methamphetamine associated cardiomyopathy
- right heart failure
- dysrhythmias
- sudden cardiac death
- infectious endocarditis
Differing patterns of methamphetamine – associated cardiomyopathy (1)
| Pattern | Presumed mechanism | Reported in literature |
|---|---|---|
| Dilated | Direct toxicity of methamphetamine to cardiac myocytes | Rajs 1979, Jacobs 1989, Nestor 1989, Hong 1991, Wijetunga 2003, Ito 2009 (6, 7,8,9,10,11) |
| Hypertrophic | Profound hypertension (increased peripheral vascular resistance) from activation of peripheral α- and β-adrenoreceptors | Nishida 2003 (5) |
| Stress cardiomyopathy (Takotsubo or reverse-Takotsubo pattern) | Acute effect of catecholamines on adrenoreceptors in myocardium | Movahed 2008, Srikanth 2008 (12,13) |
- Co- ingestion of other illicit drugs such as cocaine may increase the toxic cardiac effects either by direct additive toxic effect or drug – drug interaction leading to potentiation and prolongation of drug efficacy
- Methamphetamine cardiomyopathy refers to a non ischaemic cardiomyopathy but there is also an increase in ischaemic cardiomyopathy in patients who take methamphetamines
- Vulnerability to getting methamphetamine cardiomyopathy is genetically linked
- Abusers have consistently worse severity of disease compared to controls with non methamphetamine cardiomyopathy (11)
- Bi-ventricular cardiac pacing improves prognosis in well-treated heart failure patients with a left ventricular ejection fraction (EF) <35% and a QRS duration >120msec on their electrocardiogram (ECG).
- Automatic implantable cardiac defibrillators (AICDs) improve cardiac prognosis in patients with primary ventricular arrhythmias and those with an EF < 35% who have been stabilised on medical therapy.
Management of amphetamine induced acute hypertension
- Methamphetamine induced tachycardia and hypertension are both α and β adrenoreceptor mediated. Therefore there is a need to block both α and β adrenoreceptors
- Nitrates, benzodiazepines and α antagonists such as phentolamine are the first line antihypertensives in the acute setting. ACE inhibitors, Ca channel blockers can also be used acutely to reduce the catecholamine surge
- β antagonists used acutely and alone may cause paradoxical worsening of hypertension due to unopposed α mediated vasoconstriction.
ECHO in the diagnosis of ventricular thrombus (2,3)
- Excellent modality if good visualisation of the heart with ECHO- specificity (85 – 90%) and sensitivity (95%)
- An LV thrombus is defined as a discrete echodense mass in the LV with defined margins that are distinct from the endocardium and is seen throughout systole and diastole
- It is located adjacent to an area of the LV wall which is hypokinetic or akinetic and seen in 2 views (usually apical and short axis)
- False positives include trabeculae, papillary muscles, and side lobe artifacts
- Often the LV apex is difficult to see on TTE, so that 10 -46% of echos are inconclusive for thrombi
- IV echo contrast improves diagnostic accuracy
LV thrombus formation (2,3)
- LV thrombus formation is common post myocardial infarction, in patients with ventricular aneurysms or in patients with severe LV dysfunction
- In one study (3) the prevalence of LV thrombi was noted to be highest among patients with non ischaemic dilated cardiomyopathy (39.3%), followed by post myocardial infarction (29.8%).
- Factors causing LV thrombi in patients with non ischaemic cardiomyopathy include biventricular dilatation and severe LV dysfunction (EF <30%). Both left and right ventricles can be affected (In one study (3) 15.2% had right as well as left ventricular thrombi )
- Factors causing LV thrombi in patients following myocardial infarction include the region and extent of involvement of the infarct, the formation of LV aneurysms, and the extent of systolic dysfunction. With thrombolytics and PCI the prevalence of LV thrombi post infarction has greatly reduced (5 – 15%). Most patients affected now present with thrombi after unrecognised MI resulting in heart failure.
- Thrombi can form within 24 hours post myocardial infarction.
Teaching point: With the increase prevalence of use of methamphetamines, the Emergency Physician should be aware and looking out for cardiac complications of methamphetamine use. Look for LV thrombi in patients with any form of cardiomyopathy who are not anticoagulants. Patients who present with acute heart failure post unrecognised MI have a risk of having LV thrombi.
Bibliography
- The Cardiac Complications of Methamphetamines Elizabeth D. Paratz, MBBS a*, Neil J. Cunningham, MBBS, FACEM Andrew I. MacIsaac, MD Heart, Lung and Circulation (2016) 25, 325–332
- Education in Heart ACUTE CORONARY SYNDROMES Left ventricular thrombus formation after acute myocardial infarction
- Clinical Study Prevalence and Aetiology of Left Ventricular Thrombus in Patients Undergoing Transthoracic Echocardiography at the University of Maiduguri Teaching Hospital Mohammed Abdullahi Talle,1,2 Faruk Buba,1,2 and Charles Oladele Anjorin1 Advances in Medicine
Volume 2014 (2014), Article ID 731936, 8 pages - Islam, M.N., Kuroki, H., Hongcheng, B., Ogura, Y., Kawaguchi, N., Onishi, S. et al. Cardiac lesions and their reversibility after long term administration of methamphetamine. Forensic Sci Int. 1995; 75: 29–43
- Nishida, N., Ikeda, N., Kudo, K., and Esaki, R. Sudden unexpected death of a methamphetamine abuser with cardiopulmonary abnormalities: a case report. Med Sci Law. 2003; 43: 267–271
- Rajs, J. and Falconer, B. Cardiac lesions in intravenous drug addicts. Forensic Sci Int. 1979; 13: 193–209
- Reversible dilated cardiomyopathy induced by methamphetamine. Clin Cardiol. 1989; 12: 725–727
- Nestor, T.A., Tamamoto, W.I., Kam, T.H., and Schultz, T. Crystal methamphetamine-induced acute pulmonary edema: a case report. (460)Hawaii Med J. 1989 Nov; 48: 457–458
- Hong, R., Matsuyama, E., and Nur, K. Cardiomyopathy associated with the smoking of crystal methamphetamine. JAMA. 1991; 265: 1152–1154
- Wijetunga, M., Seto, T., Lindsay, J., and Schatz, I. Crystal methamphetamine-associated cardiomyopathy: tip of the iceberg?. J Toxicol Clin Toxicol. 2003; 41: 981–986
- Ito, H., Yeo, K.K., Wijetunga, M., Seto, T.B., Tay, K., and Schatz, I.J. A comparison of echocardiographic findings in young adults with cardiomyopathy: with and without a history of methamphetamine abuse. Clin Cardiol. 2009; 32: 18–22
- Movahed, M.R. and Mostafizi, K. Reverse or inverted left ventricular apical ballooning syndrome (reverse Takotsubo cardiomyopathy) in a young woman in the setting of amphetamine use. Echocardiography. 2008; 25: 429–432
- Srikanth, S., Barua, R., and Ambrose, J. Methamphetamine-associated acute left ventricular dysfunction: a variant of stress-induced cardiomyopathy. Cardiology. 2008; 109: 188–192





Excellent teaching & presentation